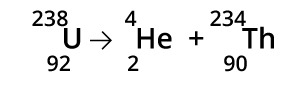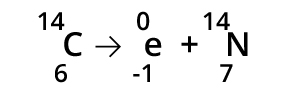Greetings! This site has migrated to riversidesci.weebly.com -- please use that site for the most current information!
Nuclear Chemistry:
|
Learning Targets:
Students will be able to...
|
RADIATION and NUCLEAR REACTIONS
Traditional chemical reactions occur when atoms bond due to their unstable number of valence electrons. However, in 1896, scientists discovered how radiation (energy) was emitted from atoms with an unstable nucleus. This created the study of Nuclear Chemistry. Nuclear chemical reactions involve changes to the particles in an atom's nucleus and therefore can cause the identity of the atom to change! All elements heavier than Bismuth (#83) exhibit natural radioactivity which can cause them to "decay" or break apart into smaller atoms. Essentially, atoms can become unstable due to a large number of protons in their nucleus (positive charges repel each other!) or an unstable ratio of protons to neutrons.
There are three common types of radiation which change an atom's nucleus:
Traditional chemical reactions occur when atoms bond due to their unstable number of valence electrons. However, in 1896, scientists discovered how radiation (energy) was emitted from atoms with an unstable nucleus. This created the study of Nuclear Chemistry. Nuclear chemical reactions involve changes to the particles in an atom's nucleus and therefore can cause the identity of the atom to change! All elements heavier than Bismuth (#83) exhibit natural radioactivity which can cause them to "decay" or break apart into smaller atoms. Essentially, atoms can become unstable due to a large number of protons in their nucleus (positive charges repel each other!) or an unstable ratio of protons to neutrons.
There are three common types of radiation which change an atom's nucleus:
|
Alpha Radiation is the emission of an alpha particle (2 protons and 2 neutrons) from an atom's nucleus. The alpha particle is essentially a Helium atom. This loss of protons and neutrons causes the element to transmutate (decay) into a lighter element. Uranium can transmutate into Thorium by emitting an alpha particle.
Beta Radiation occurs when a neutron transmutates into a proton and an electron. The electron is emitted from the nucleus but the atom's identity changes due to the additional proton. The isotope of Carbon with a total mass of 14 can decay into a Nitrogen atom through beta decay.
Gamma Radiation involves the emission of energy from an atoms nucleus. No particles are emitted so this type of radiation does not change the identity of an atom. However, gamma radiation can happen alongside alpha or beta radiation.
|
Comprehension Checkpoint 1:
TRUE or FALSE? Radiation can result in an atom having a different atomic number.
HALF-LIFE
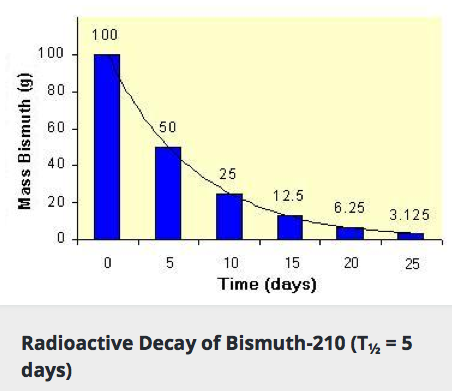
Radioactive decay occurs at a predictable rate for different elements. The rate of decay is reported as a Half-Life which is the amount of time necessary for one-half of the material to decay into a new substance. For example, if we start with a sample of 100 grams of Bismuth and only 50 grams remains in five days, then the half life is five days. And then, five days later, half of the remaining sample would decay so only 12.5 grams of Bismuth would remain. Over the course every five days, half of the remaining sample decays into Thallium through Alpha Radiation. Different types of elements have different half-lives but in general, a shorter half-life means a more unstable element because the sample is quickly decaying.
Radioactive decay occurs at a predictable rate for different elements. The rate of decay is reported as a Half-Life which is the amount of time necessary for one-half of the material to decay into a new substance. For example, if we start with a sample of 100 grams of Bismuth and only 50 grams remains in five days, then the half life is five days. And then, five days later, half of the remaining sample would decay so only 12.5 grams of Bismuth would remain. Over the course every five days, half of the remaining sample decays into Thallium through Alpha Radiation. Different types of elements have different half-lives but in general, a shorter half-life means a more unstable element because the sample is quickly decaying.
Comprehension Checkpoint 2:
An element has a half life of two days. How much of a 60 gram sample will remain after four days?
STIMULATED NUCLEAR REACTIONS
While many elements undergo radioactive decay naturally, nuclear reactions can also be forced to occur. There are two types of stimulated nuclear reactions:
While many elements undergo radioactive decay naturally, nuclear reactions can also be forced to occur. There are two types of stimulated nuclear reactions:
|
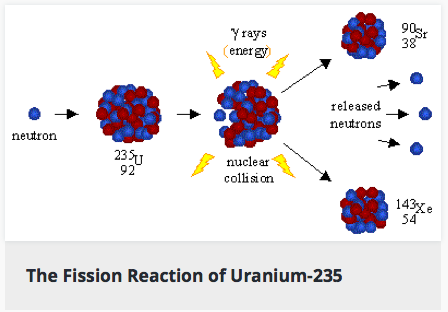
Nuclear Fission: reactions in which an atom's nucleus splits into smaller parts and releases a large amount of energy. Most commonly, this is done by "firing" a neutron at the nucleus of an atom. The energy of the neutron "bullet" causes the target element to split into two (or more) smaller atoms. The smaller atoms can then run into more of the target element and cause a chain reaction which releases even more energy (this is how nuclear bombs work).
|
|
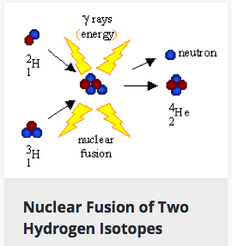
Nuclear Fusion: reactions in which two or more elements "fuse" together to form one larger element and release energy in the process. Hydrogen can fuse into the element Helium and this is how our sun emits energy! People think of our sun as a large fireball but the hydrogen gas is not burning but is undergoing nuclear fusion. Helium in the sun can then fuse into even heavier elements and this is how elements like neon, calcium, and iron were created in our Universe. No elements heavier than iron can be created in stars because the fusion reaction would require more energy than it emits.
|
Comprehension Checkpoint 3:
TRUE or FALSE: Both fusion and fission change atoms into new elements.
TRUE or FALSE: Both fusion and fission release energy.
ASSESS:
The Learning Targets listed at the top of this page will be assessed with an open notes quiz.
The Learning Targets listed at the top of this page will be assessed with an open notes quiz.